How plants decide whether to become a tree or a turnip
Written by Dr Donghwi Ko, Dr Raili Ruonal, Dr Eva Hellmann and Professor Ykä Helariutta.
Despite being one of the most economically important plant tissues, xylem is often overlooked.
In this feature, scientists from the Sainsbury Laboratory uncover its hidden complexity following their recent Science publication, revealing how a key mechanism shapes plant vascular systems and determining whether plants develop soft storage organs or the rigid woody tissue of trees.

The swelling of the turnip root is caused by the expansion of parenchyma cells, which are especially adapted to store carbohydrates.
How plants control fleshy and woody tissue growth
One of the least visible parts of a plant is also one of the most economically important – and it has nothing to do with fruit, seeds or leaves.
It is the vascular tissues that runs through stems and roots that act as the plumbing and scaffolding that determines whether a plant becomes a towering tree or a swollen, edible root.
For centuries, humans have relied on structures built from plant vascular tissue.
Without wood, our history of construction, paper-making and fuel would look very different. Without storage organs such as potatoes, cassava or turnips, global diets would also be very different.
At the heart of both timber and tubers lies the same tissue: xylem.
Xylem provides the main material that makes wood, paper, edible tubers. And of course, xylem is also responsible for making life sweeter by giving us maple syrup.
You might have briefly encountered the plant transport tissues – xylem and phloem – during secondary school or A-level Biology. Despite its importance, you are unlikely to see the word xylem appear anywhere else other than biology textbooks.

Xylem vessel cells are lignified and rigid, providing mechanical strength to timber.
The cells that shape a plant’s destiny
Xylem is made of two main cell types with very different jobs.
Vessel cells are dead, hollow tubes reinforced with secondary cell walls enriched with lignin. They act as long-distance pipelines, transporting water and minerals from roots to shoots. Because they are lignified and rigid, they also provide mechanical strength.
Parenchyma cells, by contrast, are alive. They have thin primary walls and specialise in storage, maintenance and repair. In storage organs such as radish or sugar beet, parenchyma cells dominate, swelling with starch or sugars.
In other words, the balance between vessel cells and parenchyma cells determines whether tissue becomes rigid and woody or soft and edible.
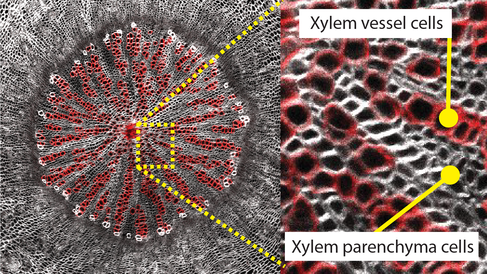
Cross section of Arabidopsis thaliana hypocotyl showing xylem tissue patterning formed by two different cell types – xylem vessels (red) and xylem parenchyma cells. SR2200 and Fuchsin staining of vessels. Imaged by Donghwi Ko.
A conserved system across plants
The evolution of plant vascular systems is remarkably conserved. Many of the genetic mechanisms that control vascular development are shared between distantly related plants. This means that discoveries in one species can often be translated to another.
Our research focuses on understanding how plants fine-tune the growth of their vascular system to prioritise transport and support or storage and expansion.
We study a small weed to understand big trees
The most recognisable vascular systems are found in trees, where a cross-section of the tree trunk reveals growth rings that indicate the age of a tree.
But trees are large and slow-growing, making them impractical for rapid genetic experiments.
Instead, plant scientists around the world turn to Arabidopsis thaliana (thale cress) to study plant development.
Arabidopsis is a small annual weed that rarely grows taller than 20cm. Despite its modest appearance, Arabidopsis has transformed our understanding of plant development.
Many of the molecular pathways that control secondary growth in trees, including hormone signals such as auxin, cytokinin, gibberellin and ethylene, and regulatory molecules involved in the signalling pathways, operate in similar ways in Arabidopsis.
Studying this miniature plant allows us to dissect mechanisms that would otherwise take years to unravel in trees.

Arabidopsis thaliana is a model plant species used to study plant development.
A mutant that changed the question
By chance, we stumbled upon a surprising mutation in Arabidopsis. Instead of forming typical roots, the mutant began producing rigid, woody structures reminiscent of trees.
We named the mutant overachiever (ovac). It produces excess vessel cells at the expense of parenchyma cells. That imbalance provided a powerful tool to explore one of the central questions in plant development: how does a cell decide whether to become a water-conducting vessel or a storage parenchyma cell?
Ribosomes and the fate of a cell
We tend to focus on DNA when we think about what is controlling what is happening in cells. However, there are tiny molecular machines in cells called ribosomes that steer cell functions by controlling when and where proteins are made. They don’t just passively read strands of genetic code, but are playing an active role in regulating cells.
By studying ovac, we uncovered a regulatory mechanism that links the protein-making machinery of the cell (the ribosome) to vascular cell fate.
Inside ribosomes lies a catalytic core known as the peptidyl transferase centre, which forms peptide bonds during protein synthesis. We found that a chemical modification (methylation) of ribosomal RNA at this centre affects how specific proteins involved in xylem development are translated.
This methylation promotes the binding of a small positively charged molecule called thermospermine. Together, these features of the ribosome alter the translation rates of different classes of developmental regulators, effectively tipping the balance between vessel formation and parenchyma development.
Our findings, published in Science, reveal that subtle changes at the level of the ribosome can reshape the architecture of plant vascular systems. This adds a new layer of control to how plants fine-tune tissue development.

Crops such as potato, cassava (pictured), sweet potato, carrot, turnip and sugar beet all rely on the proliferation of parenchyma cells capable of storing starch or sugars.
From wood to food
We identified a crucial mechanism that allows plants to shape their vascular systems, determining whether they grow soft edible storage organs or develop the rigid woody tissue characteristic of trees.
Understanding how plants fine-tune their vascular development offers a promising path for future work aimed at optimising growth traits that are critical to agriculture and forestry, including the production of commercially valuable materials such as wood, paper, edible roots and bioproducts.
The properties of wood, including strength and density, depend on the ratio and patterning of vessels, fibres and parenchyma cells. Understanding how this ratio is controlled could influence forestry, bio-based materials and carbon storage strategies.
Many staple crops form storage organs from vascular-derived tissues. Crops such as potato, cassava, sweet potato, carrot, turnip and sugar beet all rely on the proliferation of parenchyma cells capable of storing starch or sugars.
Although these storage organs differ in their precise anatomy (some arise from hypocotyls/taproots, others from roots or underground stems) they share a common feature: a high abundance of parenchyma cells.
If we can better understand how plants maintain cells in a storage state rather than a lignified vessel state, we may be able to increase tuber size or storage capacity. Insights from Arabidopsis secondary growth are particularly valuable because many storage-root crops are not yet easily amenable to advanced molecular genetics.
Choosing between strength and storage
Ultimately, plants face a developmental trade-off. Should they invest in rigid, lignified tissue to support height and water transport? Or should they invest in living storage tissue to stockpile energy?
Our work suggests that this choice is controlled not only by hormones and transcription factors but also by the translational machinery of the cell itself. By adjusting ribosomal modifications and signalling inputs, plants fine-tune the balance between vessel and parenchyma formation.
This decision has profound consequences. It influences drought resilience, wood quality, and the yield of storage crops that feed billions of people.
The next time you sit at a wooden table or peel a potato, you are seeing two outcomes of the same developmental system — a system that begins with a cellular decision inside vascular tissue: tree or turnip?
