Small amounts of a chemical hormone can reprogram a plant cell and change it’s growth and physiology. Which plant cells produce these chemicals? Where and when do these chemical hormones go? And what genetic programs do they influence once they arrive?
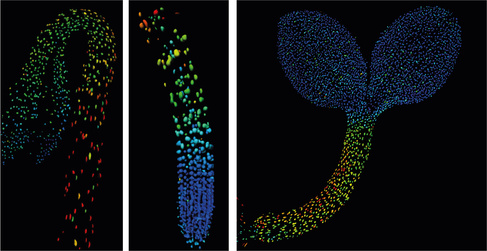
These questions are now easier to answer for one such chemical, the growth hormone gibberellin. A publication from the labs of Alexander Jones, Sainsbury Laboratory, Cambridge University and Wolf Frommer, formerly of Carnegie Plant Biology, Stanford describes the invention of a fluorescent biosensor that permits the tracking of gibberellins at the cellular level in living plants. SLCU postdocs Annalisa Rizza and Ankit Walia pursued the first lines of experimentation with the new biosensor, termed Gibberellin Perception Sensor 1 (GPS1) and found that gibberellins accumulate in rapidly elongating cell types of root tips, hypocotyls, and stamen filaments. Using GPS1 and in vivo imaging of the reference plant Arabidopsis thaliana, the team was able to observe apparent gradients of gibberellin in roots and dark-grown hypocotyls. But how are these spatial gradients set-up and maintained in these growing tissues?
The article, recently published in Nature Plants, highlights that a combination of spatially regulated biosynthesis, transport, and breakdown of gibberellins might act in concert to determine these gradients and that at least of some of these steps are regulated by a class of light responsive transcription factors. The influence of light on gibberellins and therefore growth is a classic example of plants responding to their environments. Examining hormone dynamics and growth at the cellular level during such responses is just one exciting possibility enabled by GPS1.
