The Schornack team has discovered that increasing the activity of a single gene can increase a plant’s resistance to blight at its first line of defence — the epidermis.
In the 1840s, a mysterious single-celled organism devastated Ireland's potato crops, contributing to a catastrophe, the 'Great Hunger', in which millions of people died or were forced to emigrate. We now know that the crop failures were the result of late blight disease caused by Phytophthora infestans (“Plant destroyer”), an oomycete, or water mould.

Destitution in Ireland. Failure of the Potato Crop (1846) . Image courtesy of viewsofthefamine.wordpress.com
Some might assume that this threat has been consigned to history but together with more than 100 other species of Phytophthora, this pathogen continues to wreak havoc on agriculture across the globe.
Despite huge international efforts to breed more resistant crops and develop chemical controls, Phytophthora has proven highly efficient at evolving strategies to overcome both, placing scientists and farmers in a perpetual race against the pathogen.
At the Sainsbury Laboratory in Cambridge, Sebastian Schornack’s research team recently made an important breakthrough. They have discovered that increasing the activity of a single gene can increase a plant’s resistance to blight at its first line of defence: the epidermis.

Sebastian Schornack (left), Temur Yunusov (middle) and Midhat Ubaid (right) at the Sainsbury Laboratory, University of Cambridge.
While investigating how plants and microbes interact, one of Schornack’s PhD students noticed that a plant gene called Glycerol-3-phosphate acyltransferase 6(GPAT6) – which is known to be associated with flower and seed development – became more active in plant leaves infected by the potato late blight pathogen.
Now a postdoctoral researcher at the Cambridge Institute for Medical Research, Dr Stuart Fawke recently shared this finding as the lead author of a study published in New Phytologist.
“We were looking for plant genes that were activated in response to their leaves being infected with a pathogen”, Fawke explains. “Our study is the first to link the GPAT6 gene to a plant’s immune response.”
Current disease resistance breeding in plants focuses on increasing the plant’s immunity to pathogens. This involves breeding plants with immune receptors that recognise and then kick-start the immune response against specific pathogens that dare to approach or enter the plant’s cells. But the nature of the plant’s immune system means that naturally diverse pathogens like Phytophthora eventually overcome this immunity.
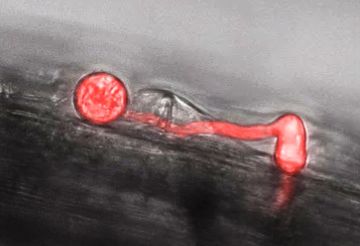
Germinating Phytophthora spore
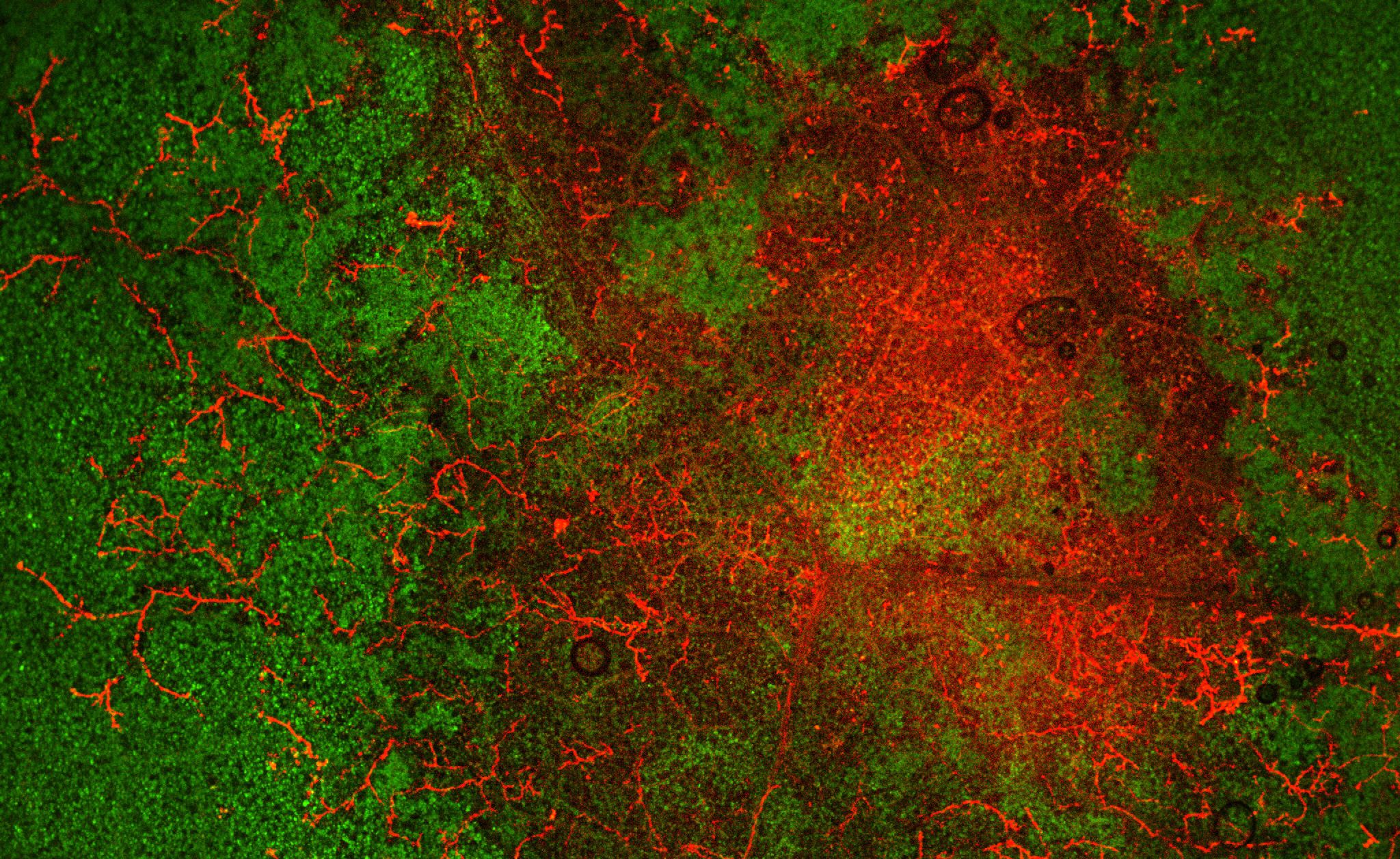
Phytophthora infestans invading Nicotiana leaf
“Receptor proteins in plants detect very specific molecular features of a pathogen or particular changes that a pathogen inflicts on a cell”, says Dr Schornack.
“Modern plant breeders aim to improve disease resistance by expanding the collection of immune receptors in new plant varieties, but this doesn’t work for long because pathogens like Phytophthora evolve new strategies to suppress or evade this resistance through natural selection.”
“An alternative to this trench warfare is to stop the pathogen at the gate.”
To test whether GPAT6 was really playing a self-defence role to fight off late blight, the team infected two plant species of the potato family, tomato and wild tobacco. The plants that had artificially heightened GPAT6 gene activity proved more resistant to the infection. By contrast, the plants with supressed GPAT6 gene activity or those that had lost the gene altogether, were more susceptible.
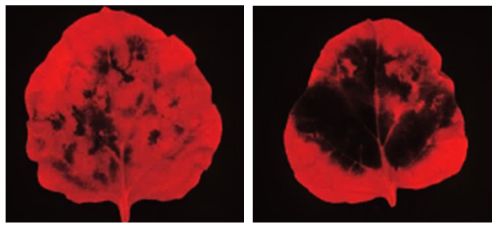 When GPAT6 activity is restricted by gene silencing (right leaf), infection is much more severe. Dark areas indicate where leaf tissue has died.
When GPAT6 activity is restricted by gene silencing (right leaf), infection is much more severe. Dark areas indicate where leaf tissue has died.
The initial physical barrier that pathogens encounter in their quest to invade plants is the epidermis, which consists of a waxy cuticle laying on top of the cell wall on the outer surface of stems and leaves: the cell wall-cuticle superstructure.
Collaborating author and plant cuticle expert, Professor Jocelyn Rose from Cornell University, found that GPAT6 was controlling some of the cuticle’s properties. He observed that in plants where GPAT6 is more active, the cuticle contains more cutin monomers (a molecule that can bond with at least two other monomer molecules) and long-chain fatty acids, which form the building blocks of the cuticle.
Thomas Torode, a postdoctoral researcher in the Schornack team, then took a closer look to see if the cuticle in plants with little to no GPAT6 activity was having an effect on the structure of the underlying cell wall. He found that low or no GPAT6 plants had an impaired cuticle and thicker, but more porous cells walls.

The cell walls of a tomato plant lacking GPAT6 (right) appear much thicker than when GPAT6 is active (left).
“We think that the lack of an intact cuticle causes the outer cell walls to swell, becoming fluffier, which allows the blight pathogen to cross”, Dr Torode says. “The imperfect cuticle also causes the plant to lose more water. The plant responds by reducing its stomata, the leaf openings that facilitate gas and water exchange, to avoid drought.”

Dr Thomas Torode at the Sainsbury Laboratory
Dr Schornack says: “Understanding how the cell wall and the plant immunity system control these invading microbes should help us to develop more resistant crops. And in doing so, we can reduce farmers’ reliance on fungicides. Resistance to fungicides is increasing and there are proposals to phase out certain chemicals like Mancozeb so our food producers need help.”
“Modifying the steps involved in building the cuticle could be used to reinforce this first-line of defence but we need to make sure that this doesn’t adversely affect other plant processes. For example, plants lacking the GPAT6 gene are more resistant to a different pathogen, grey mould (Botrytis cinerea). This is a common pathogen of strawberries and vegetables, so we need to be careful that in strengthening one shield we don’t lower another.”
“There’s no silver bullet. The pathogens will out-compete us again and again if we rely solely on traditional immunity strategies. The answer already lies within plants. For generations, we have bred plants for increased yields but too often we’ve neglected traits for protection.”
“We need to develop more efficient cropping systems to meet the growing global demand for food and anything that increases disease resistance by using the plant’s own defence strategies, rather than chemicals, is a step in the right direction.”
Reference:
Fawke, S., Torode, T.A., Gogleva, A., Fich, E.A., Sørensen, I. , Yunusov, T., Rose, J.K.C. & Schornack, S., 'Glycerol phosphate acyltransferase 6 controls filamentous pathogen interactions and cell wall properties of the tomato and Nicotiana benthamiana leaf epidermis.' New Phytologist (2019).
Acknowledgements:
This research was funded by the Royal Society and the Gatsby Charitable Foundation.
This article was written by Tom Almeroth-Williams & Kathy Grube and originally published on the University of Cambridge website. Read the original article.
