Research Interests
Plasticity in plant growth
Throughout my scientific career, my research has revolved around one key question — how do plants grow and respond to intrinsic signals such as hormones or extrinsic environmental cues such as light or temperature? Plants need to constantly adjust their growth according to their immediate environment not only for survival but for optimisation of light capture, heat dissipation, or nutrient or water uptake. One way plants achieve this is through plasticity in their developmental program and thus their morphology. Since plant cells are immobile, unlike animal cells, this plasticity is achieved at the local cell level via changes in cell division or expansion processes. I am interested in understanding how such changes in an individual or a group of cells influence the growth of entire an organ or plant.
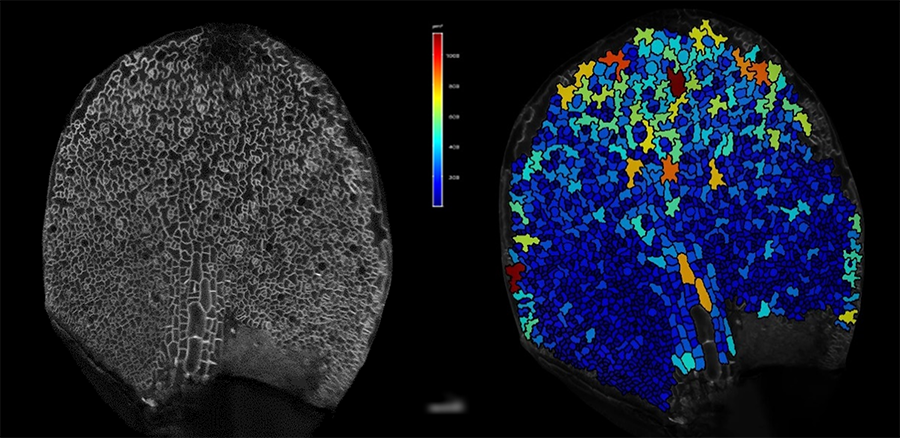
Confocal image of a young developing Arabidopsis leaf (left) showing a gradient of divinding and expanding cells along the promixal-distal and medio-lateral growth axis. Cells were color coded according their area in MorphoGraphX (right).
Biomechanics of cell division and expansion
Currently, in the Robinson group, I am taking this question further and studying how mechanical signals influence a cell’s decision to divide or progress from division to expansion. One of the ways I am approaching this is by comparing the growth of cells under ambient and high ambient temperatures (5-7 °C higher than the optimal growth temperature). Warm temperatures are known to promote cell elongation in cells in petiole and hypocotyls and inhibit cell division in the leaves of the Arabidopsis plant (Saini et. al, preprint). The focus of my research is the biomechanics of trade-offs between division and expansion utilising primarily the developmental pathway of pavement cells (epidermal cells of plants that grow in jigsaw-like shapes) in the leaves of Arabidopsis thaliana.
Research background
Integrating developmental and environmental signals to adapt growth
During my postdoc at the National Institute of Plant Genome Research, Delhi, India in Dr Aashish Ranjan’s lab, I studied how developmental signals integrate with environmental signals in bringing growth adaptations in plants. The lab approached it from an eco-evo-devo aspect and used comparative transcriptomics analysis to study the effects of shade and high temperature on tissue-specific growth changes in Arabidopsis, tomato, and rice. The RNA sequencing libraries were made in-house after standardisation of the protocol in the lab adapted from the BrAD-Seq technique mentioned in Townsley and Sinha (2015).
This work was featured in the web seminar series by Plantae Presents from ASPB (video below).
High temperature effect on leaf development
Based on phenotyping and preliminary results from RNAseq, I became fascinated by the cellular and molecular responses of leaves exposed to high temperature and showed that the high-temperature-grown leaves were smaller and produced fewer cells mainly due to suppression of the core cell division process by the bHLH transcription factor Phytochrome Interacting Factor 4, PIF4, and TCP family transcription factor, TCP4.
Auxin-regulation of leaf growth
I originally became interested in studying organ growth regulation during my Ph.D. in Kris Vissenberg’s Lab at the University of Antwerp, Belgium. I was part of a project, ''A Systems Biology Approach of Leaf Morphogenesis'', where I focussed on auxin-regulation of leaf growth in Arabidopsis. While auxin is a plant hormone known to regulate virtually every aspect of plant growth and development, I studied how changes in auxin efflux protein (PIN) polarity influenced changes in auxin concentration locally and between source and sink tissues and its effect on cell growth in the leaves.
Publications
Saini K, Dwivedi A, and Ranjan A. PIF4 and TCP4 regulate cell division under elevated temperature in Arabidopsis thaliana leaves (2022). bioRxiv https://doi.org/10.1101/2022.02.02.478810
Jathar, V., Saini, K., Chauhan, A., Rani, R., Ichihashi, Y. and Ranjan, A. (2022), Spatial control of cell division by GA-OsGRF7/8 module in a leaf explaining the leaf length variation between cultivated and wild rice. New Phytol. https://doi.org/10.1111/nph.18029
Irulappan, V., Kandpal, M., Saini, K., Rai, A., Ranjan, A., Sinharoy, S., Muthappa, S-K. (2022) Drought stress exacerbates fungal colonization and endodermal invasion and dampens defense responses to increase dry root rot in chickpea. MPMI. https://doi.org/10.1094/MPMI-07-21-0195-FI
Saini K, Markakis MN, Zdanio M, Balcerowicz DM, Beeckman T, De Veylder L, Prinsen E, Beemster GTS and Vissenberg K (2017) Alteration in Auxin Homeostasis and Signaling by Overexpression Of PINOID Kinase Causes Leaf Growth Defects in Arabidopsis thaliana. Front. Plant Sci. 8:1009. doi: 10.3389/fpls.2017.01009
Saini, K., AbdElgawad, H., Markakis, M.N., Schoenaers, S,. Asard, H., Prinsen, E., Beemster, G., Vissenberg, K. (2017) Perturbation of auxin homeostasis and signaling by PINOID overexpression induces stress responses in Arabidopsis. Front. Plant Sci. 8:1308. doi: 10.3389/fpls.2017.01308
Kalve, S*., Saini, K*., Vissenberg, K., Beeckman, T. and Beemster, G. T. (2015) Transverse Sectioning of Arabidopsis thaliana Leaves Using Resin Embedding. Bio-protocol 5(18): e1592. http://www.bio-protocol.org/e1592 (*Equal contribution)
Markakis MN, Boron AK, Van Loock B, Saini K, Cirera S, et al. (2013) Characterization of a Small Auxin-Up RNA (SAUR)-Like Gene Involved in Arabidopsis thaliana Development. PLoS ONE 8 (11): e82596. doi:10.1371/journal.pone.0082596